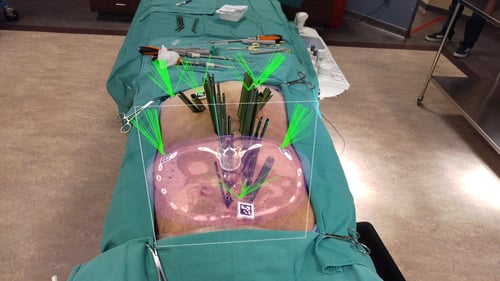
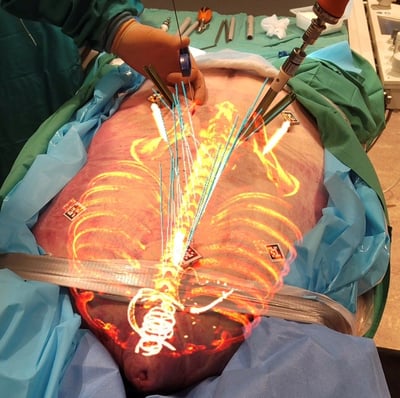
"This will immediately change how we perform surgery because it can rapidly decrease the amount of time in the OR and get the patient off the table faster with confidence that screws are placed accurately."
- Dr. Ted Andrews, Spine Surgeon, University of Pittsburgh Medical Center

.jpeg)
-1.jpg?width=500&height=325&name=ezgif.com-gif-maker%20(1)-1.jpg)
.webp)